 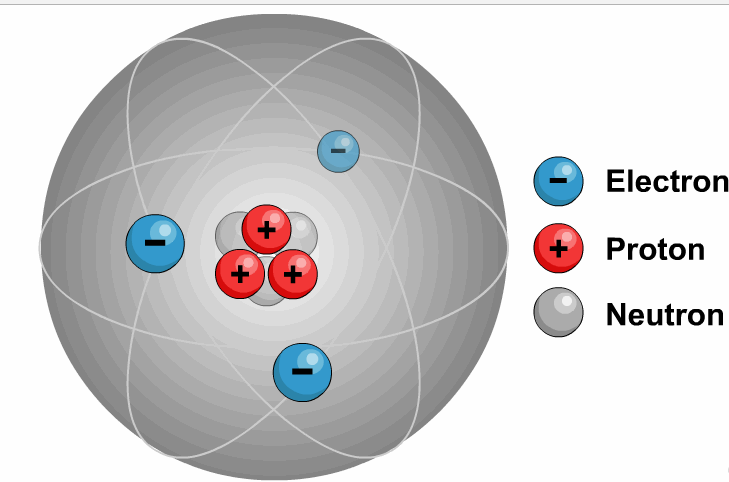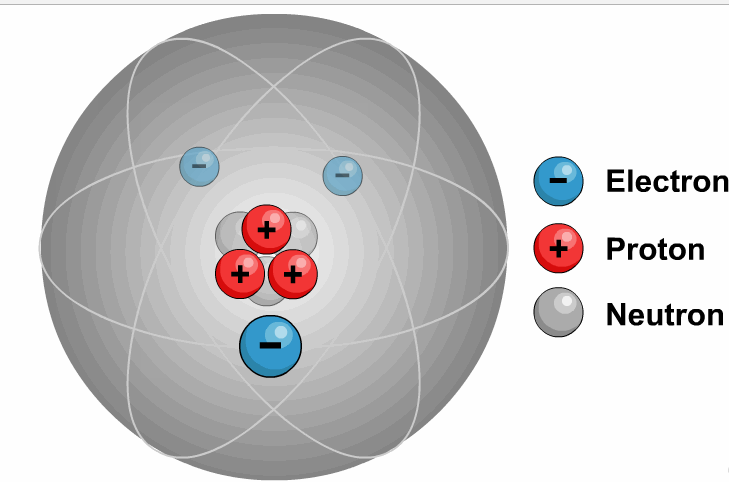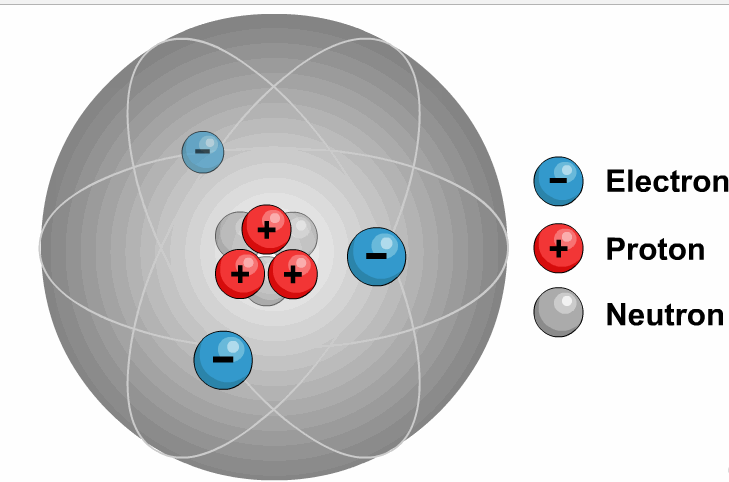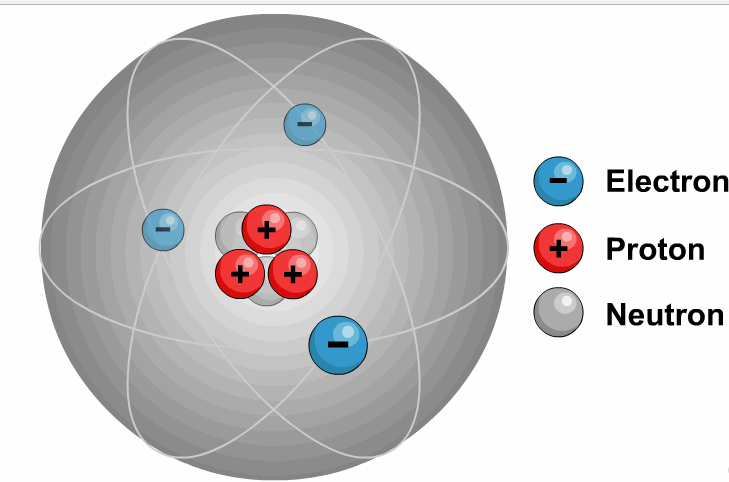
It is necessary to write the chemical formula of an atom before representing it in a charged ion form. Later, in 1884, Svante August Arrhenius further proposed the mechanism of ions in his experiment. He described ions as electrically charged atoms or a family of atoms traveling toward a cathode or anode. The word ion is acquired from the Greek work ἰόν, which means ‘to go.’ Therefore, the term ‘ion’ implies ‘a goer.’ In 1830, ions were first introduced by Micheal Faraday. If two or more different atoms form an ion, they are called molecular or polyatomic ions. If an ion is formed by only one variety of atoms (each holding some gross charge, either positive or negative), in that case, the group of atoms can be referred to as a monatomic ion or an atomic ion. Sodium ion (Na⁺), magnesium ion (Mg²⁺), chloride ion (Cl⁻), and oxide ion (O²⁻). Therefore, it contains an unequal number of electrons and protons. Ions DefinitionĪn ion is formed by an atom’s loss or gain of electrons. In simple words, what is ion or ions definition can be described as an electrically charged atom or group of atoms. As a universally acknowledged fact, ions of opposite charges always attract each other. The atoms that tend to form cations are generally known as metals, while those that tend to form anions are called non-metals. An ion with a total positive charge is termed a proton, while an ion with a net negative charge is termed an electron.Ī proton is also known as a cation, whereas you can call an electron an anion. It is an atom, group of atoms or any subatomic particle with a net electric charge. Lithium-ion batteries power everything from your mobile phone to your laptop.Ions are the chemically charged species that contain either a positive or a negatively charged quantity. Negative ions in the body relax you and energize your body. Ions are sources of energy, whether in the human body or electrical gadgets. Some examples of molecular ions are NH 4 + (ammonium), OH – (hydroxide), etc. Similar to an atomic ion, a molecular ion may be molecular cation or anion. When a molecule (a group of atoms bonded together) combines with an atomic ion, the molecule gets charged and becomes an ion. So atoms find it easier to lose or gain electrons to become ions. Protons are 1836 times heavier than electrons. Atom can gain of proton to get positively charged.Atom can lose a proton to get negatively charged.Atom can gain an electron to get negatively charged.Atom can lose an electron to get positively charged.There are 4 ways in which an atom can become an ion: This negatively charged ion is called anion. When an atom has more electrons than protons, it gets negatively charged. This positively charged ion is called cation. When an atom has more protons than neutrons, it gets positively charged. When the number of these charged particles is the same, the atom is neutral. So atoms are made up of charged particles (and also neutral ones). The protons and neutrons form the nucleus of the atom whereas electrons orbit around the nucleus, as shown in Figure 1.įigure 1: Electrons Revolving Round the Nucleus they have neither negative nor positive charge. Electrons are negatively charged while protons are positively charged. 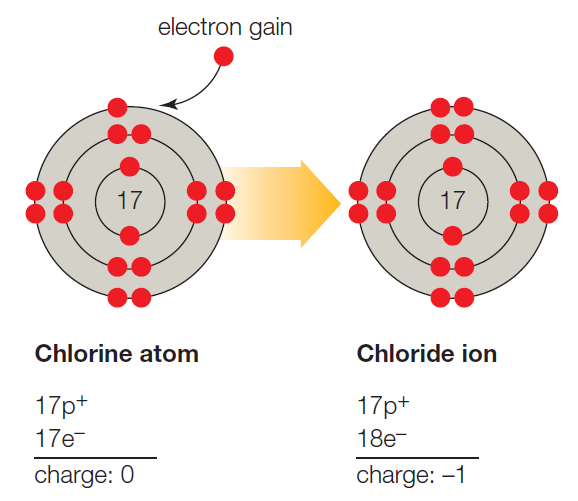
Sub-atomic particlesĪtom is made up of electrons, protons and neutrons. Alternately, we can say that atom is the smallest unit of matter that can exist in a stable form. As we discussed in a previous post, all matter is made up of atoms. Ions are nothing but atoms or molecules with positive or negative electrical charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
